International Cement Review, October 2019.
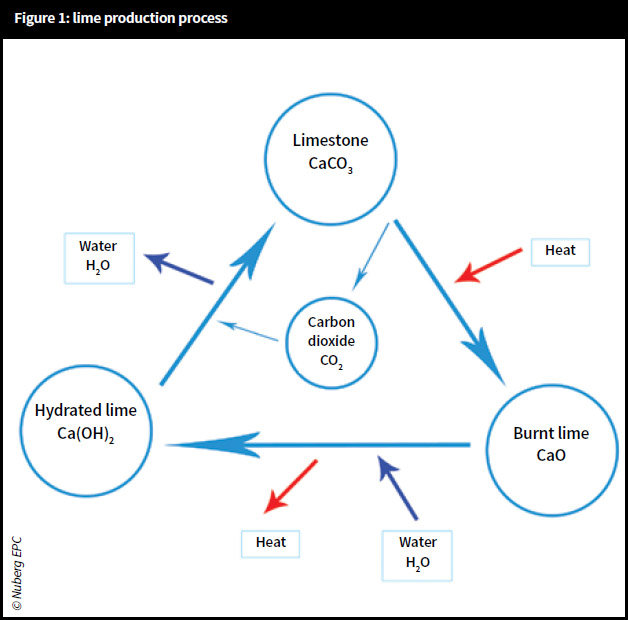
Lime is produced from limestone, which is a high-volume, low-cost commodity mineral and mainly used as a chemical additive. It is primarily alkaline and consists of several compounds of calcium. The process by which raw limestone forms lime is called a lime cycle.
There are two main types of lime available in the market: quicklime and hydrated lime. Both are used for various applications, including cement manufacturing, metal processing, agrochemicals, and pulp and paper processing. In addition, lime is used in industries within the chemical, metallurgical, construction, environmental
and agricultural sectors. In steel manufacturing, it is used as a flux to remove impurities such as sulphur, silica and phosphorus. Lime is one of the key raw
materials used in construction because its anti-stripping and binding properties are highly sought after in this industry
Global demand and main consumers for lime
According to Persistence market research, the estimated value of the global lime market was US$40.6bn in 2018 and is expected to reach US$50bn by the end of 2026, expanding at a CAGR of 2.5 percent over the forecast period. The global lime market is expected to represent an incremental opportunity of US$9bn between 2018-26.
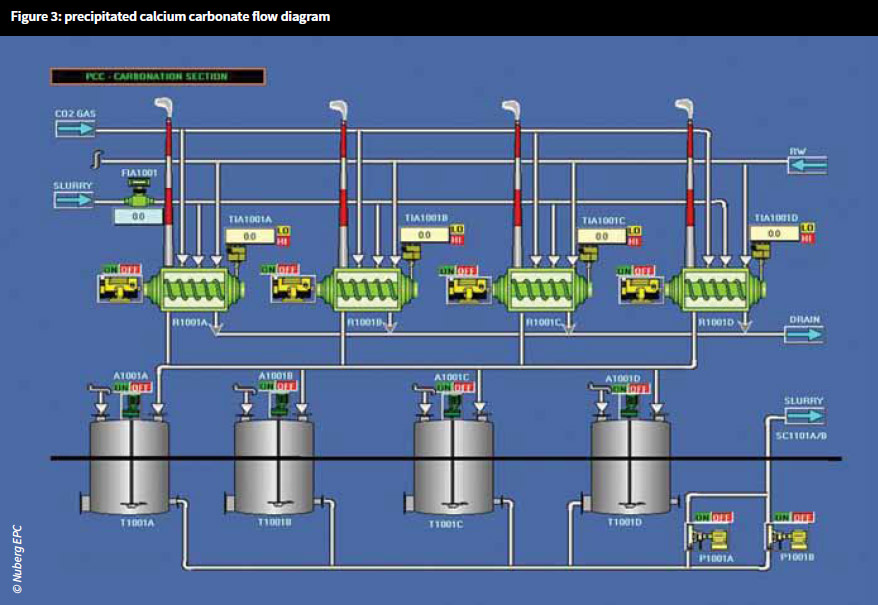
Increasing demand for mining and metallurgical applications, especially for the removal of impurities, is an important factor in the growth of the lime market across the world. In addition, the increasing use of lime in the manufacture of precipitated calcium carbonate also adds to lime demand. Precipitated calcium carbonate, due to its high calcium content, it is widely used in the production of paper, plastics, paints, rubber, calcium tablets, and multi-vitamin/mineral tablets. The use of precipitated calcium carbonate, especially in polymer, paper and healthcare industries, supports the growth of the lime market.
On a geographical level, the Asia-Pacific region dominates the lime market in terms of production and demand, followed by Europe, which together with North America had a 23 per cent share of the lime market. The key consumers of lime are China, India and Japan, with China and India accounting for around 68 per cent of global lime production in 2016. In China the lime market has witnessed steady growth over the study period, mainly due to the growing mineral production and rising government and private sector investment into infrastructure development. India’s lime market is estimated to register a healthy CAGR over the study period.
Lime production process
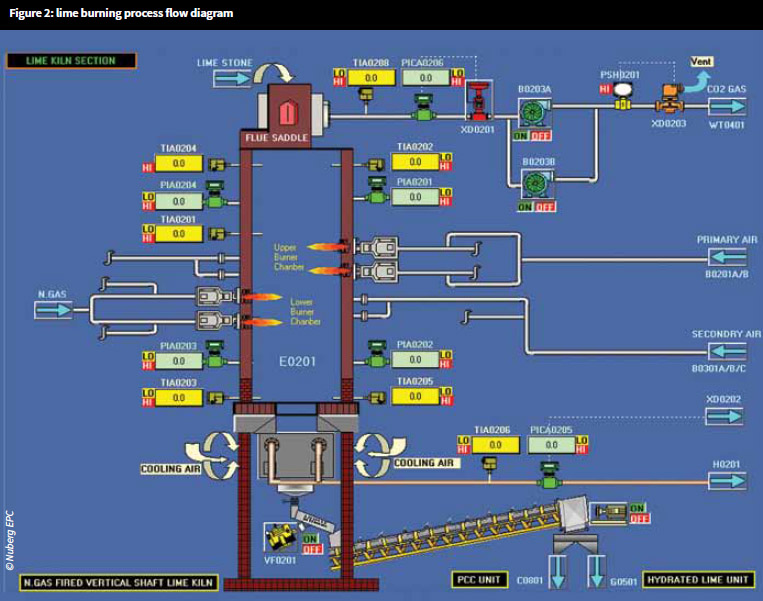
Lime burning
Limestone (calcium carbonate – CaCO3) is burnt in a kiln to produce calcium oxide (CaO) and emits carbon dioxide (CO2) gas, which is commonly known as quicklime or lumplime.
To ensure good-quality material is produced, the limestone needs to be burnt at 900°C. In addition, the temperature at which it is burnt will affect its reactivity in all other stages of the lifecycle – slaking and carbonating. At this stage, the resulting lime is at its most eruptive and dangerous form.
Lime slaking
The quicklime is then combined with water (slaked) as quickly as possible. From the moment it is burnt the material starts to degrade by air-slaking. By combining quicklime (CaO) and water (H2O), calcium hydroxide (Ca(OH)2) is produced.
There are three main ways of slaking the quicklime: in an excess of water to produce a putty, in a shortfall of water to produce powder – hydrated lime, and in damp sand to produce a hotmix.
Lime carbonation
Carbonation is the process in which lime sets by absorbing water-soluble CO2 from the air. The ‘set’ or carbonation must occur slowly – the slower the set is carried out, the better (it is not a case of just drying). Therefore, direct heaters may cause failures and it is important that the conditions are right to enable the water-borne CO2 to be absorbed. In Nuberg’s Lime Handbook specific conditions are described to control the carbonation process. Failure to properly control carbonation will lead to issues and potential failure on site.
Safety in lime production
The production of lime is subject to several safety measures, including:
- The calcining kiln should be operated carefully as it may be the cause of production ignition or burn injury.
- Keep clear of the kiln sight cap. Carbon oxide, carbon dioxide may cause poisoning.
- Lime dust is hazardous to mucous membranes and wet skin.
- To eliminate any dusting, the transport and bins should be covered with casings, caps, de-dusting devices should be installed and hydraulic suppression of dust should be applied.
- Belt conveyors should be equipped with canopies over their full length
- Operators should wear face masks and special clothes.
Lime storage and transportation
Lime is transported in roofed transport of any type. Different lime types should be stored and transported separately without any exposure to moisture or contamination by foreign substances. The guaranteed storage life is 30 days from the date of its shipment to the consumer. If stored properly, non-slaked lime preserves its properties for 15-18 days.Following its brief, the Nuberg EPC team applied its expertise to the key steps of the lime plant project.
Mode of supply
Quicklime is supplied in bulk only, while hydrated lime is supplied either in multiwall paper bags of 25kg nominal capacity or in bulk.
Ensuring the right weight
If lime is supplied in bulk, the weight of each delivery vehicle will be certified on a registered public weighbridge. A copy of each certified weighbridge docket is then included in the quality records and sent to the administrator.
If lime is supplied in bags, each bag will have the manufacturer’s certified nominal weight clearly displayed.
Rate of delivery
Lime will be delivered at the rate stated in the sales contract, except where the administrator provides a modification to this rate, mutually agreed between the administrator and the contractor.
Mode of delivery
Bulk lime deliveries will be carried out in air-tight, waterproof and water-tight blower-type vehicles. Furthermore, the delivery vehicles will be designed to enable a minimum discharge rate of 40tph into the spreader or bulk storage bin.
During the transfer, delivery or storage of the lime, the equipment needs to be waterproof, emptied, cleaned and dried prior to the introduction of each different type or source of lime.
All bagged lime deliveries will be made in air-tight, waterproof and water-tight bags, which are labelled, stacked and wrapped or strapped on pallets.
Sustainability concerns
Like many other limestone-based processes, quicklime production includes thermal decomposition of minerals in the limestone followed by the emission of gaseous CO2 to the atmosphere.
If the fuel used to generate the heat includes carbon, additional CO2 will be released. Biofuels are considered carbonneutral and excluded from emissions trade legislation (EU, 2003). While there is an increase in the use of non-fossil fuels in quicklime and cement clinker production, fossil fuels remain the main fuel used (Schorcht, 2013). The concentration of CO2, the main GHG in the atmosphere, is currently increasing at 2.1 ppm/a (Feldman, 2015). Climate modelling indicates that CO2 mitigation is not substitutable and should be prioritised (Strassmann, 2009).
It has been estimated that in the short term ~40-50 per cent of the anthropogenic CO2 remains in the atmosphere, while the rest is stored in the oceans and terrestrial biosphere (Sabine, 2004).
In the longer term, more than 90 per cent of the CO2 is expected to be dissolved in the oceans, contributing to acidification (Archer, 1998). There has been at present no full-scale CO2 abatement technology deployed in quicklime production.
References
1. EU (2003) European Union, DIRECTIVE 2003/87/ EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 13 October 2003 establishing a scheme for greenhouse gas emission allowance
trading within the Community and amending Council Directive 96/61/EC
2. Feldman, DR, Collins, WD, Gero, PJ, Torn, MS, Mlawer , EJ and Shippert , TR (2015) ‘Observational determination of surface radiative forcing by CO2 from 2000 to 2010’ in: Nature, 519 (7543), p339-343.
3. Sabine, C, Feely, RA, Gruber, N, Key, R, Lee,
K, Bull ist er, JL, Wanninkhof , R, Wong,
CS, Wall ace, DWR, Tilbrook , B, Mill ero ,
FJ, Peng, T-H, Kozyr, A, Ono, T and Rios , AF
(2004) ‘The Oceanic Sink for Anthropogenic CO2’
in: Science, 305 (5682), p367-371.
4. Schorc ht, F, Kourt i, I, Scalet, BM,
Roudier, S and Sancho, LG (2013) Best
Available Techniques (BAT) Reference Document
for the Production of Cement, Lime and
Magnesium Oxide, Industrial Emissions Directive
2010/75/EU, Integrated Pollution Prevention and
control. Luxembourg: Publications Office of the
European Union, https://eippcb.jrc.ec.europa.
eu/reference/BREF/CLM_Published_def.pdf
(Accessed 2015-09-15)
5. Str assm ann, K, Platt ner, G-K and Joos ,
F (2009) ‘CO2 and non-CO2 radiative forcings
in climate projections for twenty-first century
mitigation scenarios’ in: Climate Dynamics, 33
(6), p737-749.
6. Arc her, D, Haroo n, K and Er nst , M-R (1998)
‘Dynamics of fossil fuel CO2 neutralisation by
marine CaCO3’ in: Global Biogeochemical Cycles,
12 (2), p259-276.
This article was first published in International Cement Review in October 2019.
Media Gallery
SiliconIndia - Nuberg EPC: Where Ideas Escalate to Expanses of Engineering Excellence
World Fertilizer - A World of Sulfur
International Cement Review - A new lime plant for Crescent
Process Worldwide - Making Ideas Happen, from Concept to Commissioning
Industrial Products Finder (IPF Online) - I see a great future for EPC companies from India
Refining & Petrochemicals - Top 30 EPC Contractors
World of Chemicals - EPC demand is rising in all dimensions of chemical industry
Financial Express - Indian firms can be world leaders in EPC
The Sunday Guardian - Engineering companies enthused by Make in India
Download PDF